-
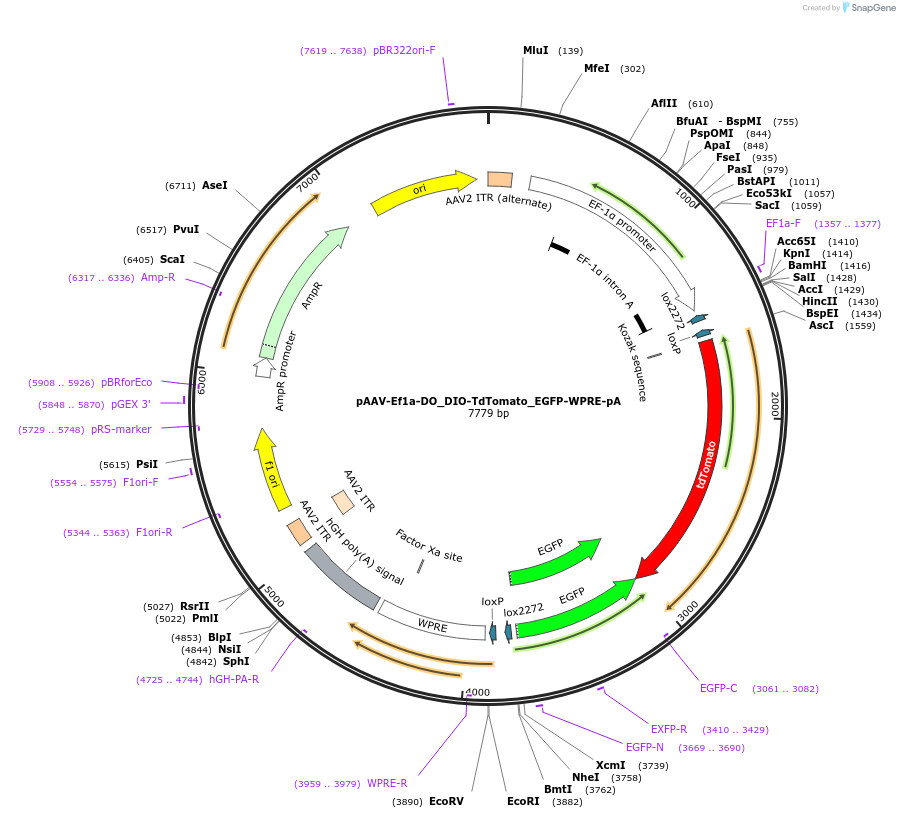
PurposeDO/DIO Cre-Switch. Expresses tdTomato in Cre negative cells, expresses EGFP in Cre positive cells
-
Depositing Lab
-
Sequence Information
Ordering
| Item | Catalog # | Description | Quantity | Price (USD) | |
|---|---|---|---|---|---|
| Plasmid | 37120 | Standard format: Plasmid sent in bacteria as agar stab | 1 | $94 | |
Backbone
-
Vector backbonepAAV-Ef1a-DIO-hChR2(H134R)-mCherry-WPRE-pA
-
Backbone manufacturerK. Deisseroth Lab
- Backbone size w/o insert (bp) 5627
- Total vector size (bp) 7778
-
Vector typeAAV, Cre/Lox ; Cre-Switch
Growth in Bacteria
-
Bacterial Resistance(s)Ampicillin, 100 μg/mL
-
Growth Temperature37°C
-
Growth Strain(s)Stbl3
-
Copy numberUnknown
Gene/Insert
-
Gene/Insert nameTdTomato-EGFP
-
SpeciesSynthetic
-
Insert Size (bp)2151
- Promoter EF1a
Cloning Information
- Cloning method Restriction Enzyme
- 5′ cloning site Asc1 (not destroyed)
- 3′ cloning site Nhe1 (not destroyed)
- 5′ sequencing primer CTTCCATTTCAGGTGTCGTG
- 3′ sequencing primer GCAGCGTATCCACATAGCG
- (Common Sequencing Primers)
Resource Information
-
A portion of this plasmid was derived from a plasmid made byKarl Deisseroth Lab, Stanford Univ.
-
Articles Citing this Plasmid
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Trademarks:
- Zeocin® is an InvivoGen trademark.
These plasmids were created by your colleagues. Please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
-
For your Materials & Methods section:
pAAV-Ef1a-DO_DIO-TdTomato_EGFP-WPRE-pA was a gift from Bernardo Sabatini (Addgene plasmid # 37120 ; http://n2t.net/addgene:37120 ; RRID:Addgene_37120) -
For your References section:
Novel recombinant adeno-associated viruses for Cre activated and inactivated transgene expression in neurons. Saunders A, Johnson CA, Sabatini BL. Front Neural Circuits. 2012 Jul 27;6:47. doi: 10.3389/fncir.2012.00047. eCollection 2012. 10.3389/fncir.2012.00047 PubMed 22866029