Viral Vector Guides and Plasmids
Viral vectors are popular tools for delivering genetic information to cells. These vectors rely on production plasmids that have been carefully optimized for both safety and efficiency. Learn more about the different types of viral vectors and find viral production and transfer plasmids for a wide array of research purposes.
Interested in Addgene's viral services? See Addgene's Packaged on Request service for made-to-order AAV vectors from Addgene's AAV plasmid collection. Addgene also has a selection of in-stock viral preps available.
Browse Guides and Plasmids
- Lentiviral Vector Guide | Lentiviral Plasmids
- Lentiviral vectors are versatile tools because of their ability to transduce dividing and non-dividing cells. These virusal vectors enable long-term stable gene expression and are permanently integrated into the host genome.
- AAV Vector Guide | AAV Plasmids
- Adeno-associated viral (AAV) vectors are commonly used for gene delivery in vivo because of their mild immunogencity. These viral vectors can direct long-term transgene expression, but generally don't integrate into the host genome. AAVs are limited by their small packaging capacity.
- Gamma-retroviral Vector Guide | Gamma-retroviral Plasmids
- Gamma-retroviral vectors, often referred to as just retroviral vectors, have a relatively high transduction efficiency and only infect dividing cells. They integrate permanently into the host genome, enabling long-term transgene expression. Retroviral vectors are popular for making stable cell lines or clinical use.
- Adenoviral Vector Guide | Adenoviral Plasmids
- Adenoviral vectors have high packaging capacity, high transduction efficiency, and high but transient transgene expression.They can induce a strong immunogenic response, useful for clinical applications as vaccines.
Selecting a Viral Vector Type
Each viral vector type exhibits different properties and are suited for specific research goals. For example, AAV vectors are typically preferred for in vivo studies because of their low immunogenicity. Viral vectors also vary in the composition of their viral genome, expression of the transgene, packaging capacity, and size.
| Vector Type | Expression | Genome | Packaging Capacity | Virus Size | Cells Infected | Host Genome Integration | Immune Response |
|---|---|---|---|---|---|---|---|
| Lentiviral | Stable | RNA | < 8 kb | 80–130 nm | Dividing/Non-dividing | Yes | Low |
| Gamma-retroviral | Stable | RNA | < 8 kb | 80–130 nm | Dividing | Yes | Moderate |
| AAV | Transient or Stable* | Single-stranded linear DNA | ∼4.5 kb | 18–26 nm | Dividing/Non-dividing | No* | Very Low |
| Adenoviral | Transient | Double-stranded linear DNA | 5–36 kb | 105 nm | Dividing/Non-dividing | No | High |
*Recombinant AAV has a low frequency of target cell genome integration.
Introduction to Viral Vectors
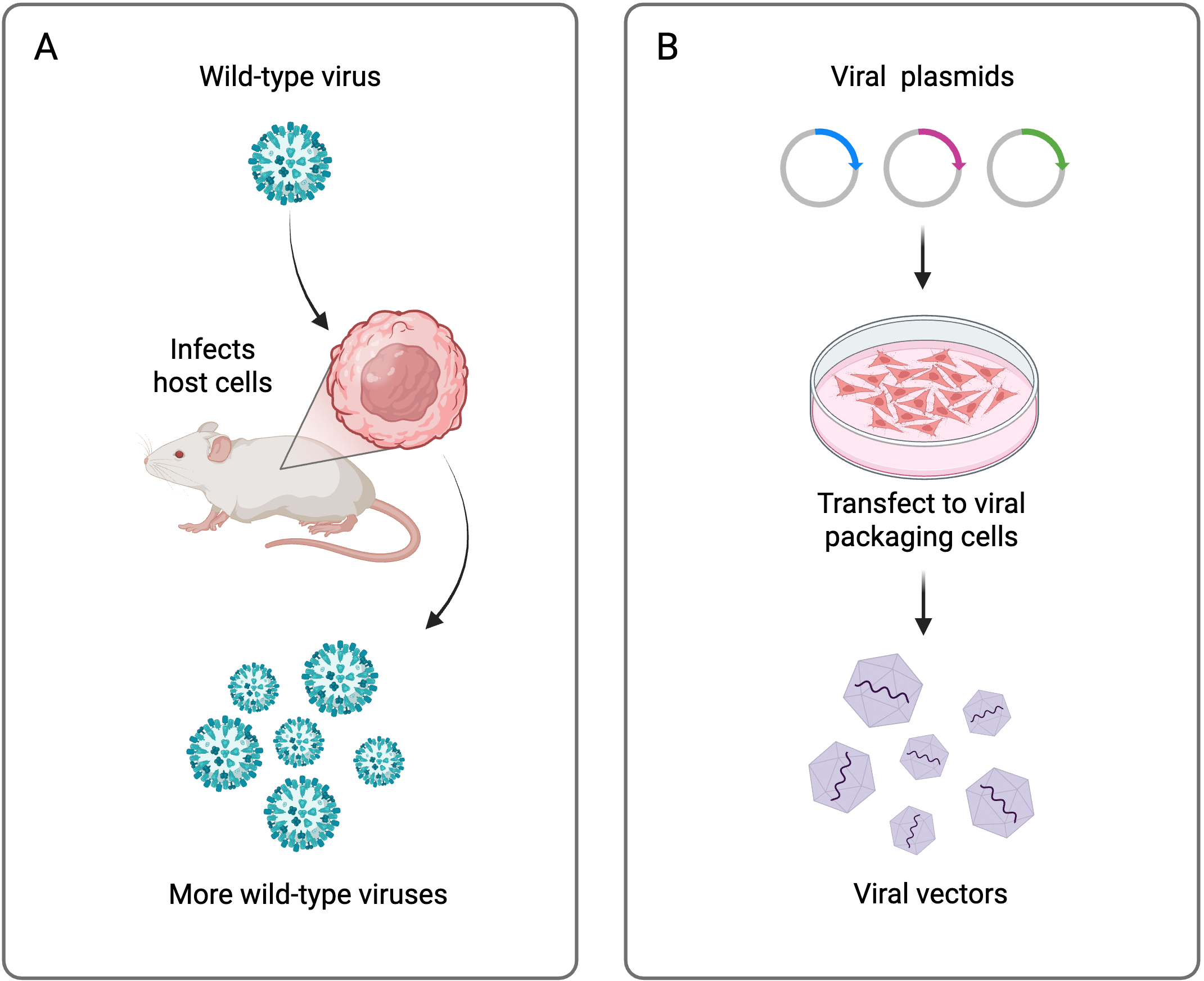
Wild-type viruses have one goal, which is to make more viruses. The viral genome contains only the necessary genes to achieve this goal. When a virus recognizes a host cell, it injects its viral genome into the cytoplasm and hijacks the host’s machinery to produce more viruses. In this way, viruses are excellent vehicles to deliver genetic cargo into cells.
To create safe and effective viral vectors, wild-type viral genomes have been stripped of any unnecessary accessory genes and divided into multi-plasmid systems. These systems consist of a plasmid containing the genetic information to be delivered (transfer or shuttle plasmid) and one or more plasmids containing the necessary components of the viral genome to assemble the viral capsids (packaging and envelope plasmids). When transfected into in vitro cell cultures, these plasmids direct the production of viral vectors that contain the desired genetic cargo. In turn, these viral vectors are used to transduce cells of interest. Specific modifications to these plasmids, such as different promoters or tags, can even direct delivery of the genetic cargo to specific cell types and tissues.
Viral plasmids have been carefully engineered for safety and are replication-incompetent. Specific genes have been mutated or deleted to prevent uncontrolled viral replication, and the required genes are divided in the multi-plasmid system. Many viral plasmids are also self-inactivating, meaning that they can only complete one round of transcription of their viral genome. Therefore, viruses used in research typically do not have the ability to propagate in the same way that wild-type viruses do.
For general virus biosafety guidelines, check out Addgene's Biosafety Resource Guide.
Resources
Content last reviewed: 5 August 2025


