-
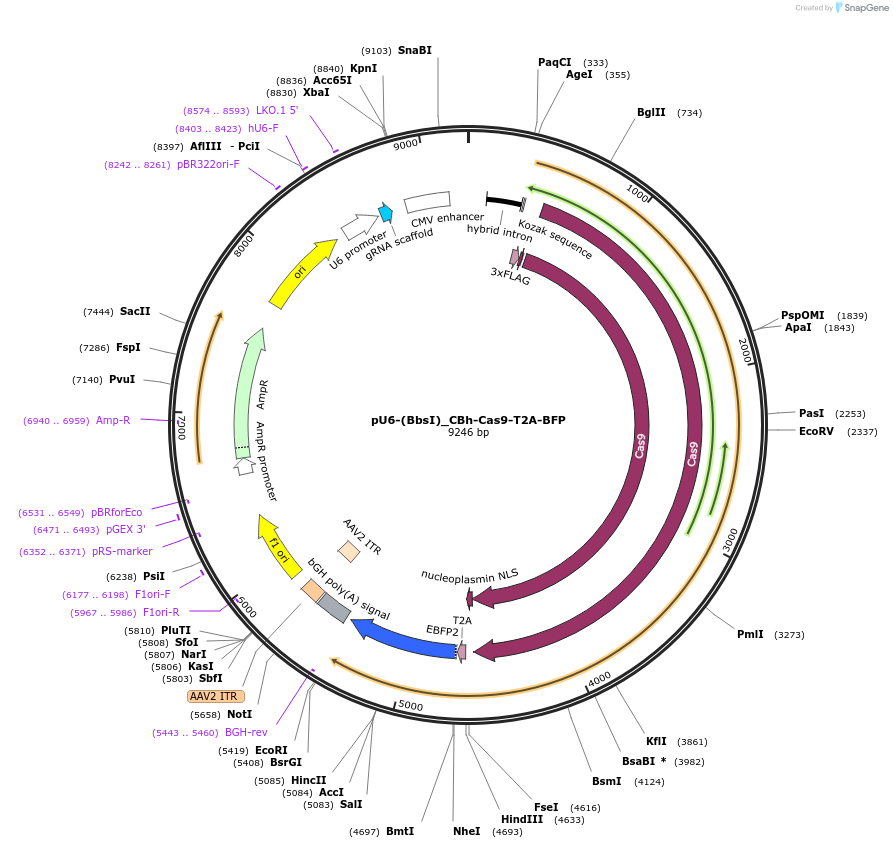
PurposeExpression vector for sgRNAs cloned into the BbsI sites and for expression of Cas9 linked to BFP via a T2A peptide
-
Depositing Lab
-
Sequence Information
Ordering
| Item | Catalog # | Description | Quantity | Price (USD) | |
|---|---|---|---|---|---|
| Plasmid | 64323 | Standard format: Plasmid sent in bacteria as agar stab | 1 | $94 | |
Backbone
-
Vector backbonepX330
-
Backbone manufacturerZhang lab (Addgene plasmid # 42230)
-
Modifications to backboneThe T2A-BFP fragment was cloned into FseI & EcoRI sites of pX330 (Addgene plasmid # 42230). The BFP template was derived from MSCV-IRES-BFP, a kind gift of Frank Rosenbauer and Martin Janz (Charite, Berlin)
-
Vector typeMammalian Expression, CRISPR
Growth in Bacteria
-
Bacterial Resistance(s)Ampicillin, 100 μg/mL
-
Growth Temperature37°C
-
Growth Strain(s)DH5alpha
-
Copy numberHigh Copy
Gene/Insert 1
-
Gene/Insert nameCas9
-
Alt name3xFLAG-NLS-Cas9-NLS-T2A-EBFP2
-
SpeciesStreptococcus pyogenes
-
Insert Size (bp)4500
-
GenBank IDNC_002737.1
- Promoter CBh
-
Tags
/ Fusion Proteins
- 3xFLAG (N terminal on insert)
- NLS (N terminal on insert)
- NLS (C terminal on insert)
- T2A-EBFP2 (C terminal on insert)
Cloning Information for Gene/Insert 1
- Cloning method Unknown
- 5′ sequencing primer pCBhPro-F (5'-agggatggttggttggtggg-3') for Cas9
- (Common Sequencing Primers)
Gene/Insert 2
-
Gene/Insert namesgRNA cassette
- Promoter U6
Cloning Information for Gene/Insert 2
- Cloning method Restriction Enzyme
- 5′ cloning site BbsI (unknown if destroyed)
- 3′ cloning site BbsI (unknown if destroyed)
- 5′ sequencing primer hU6-F (5'-GAGGGCCTATTTCCCATGATT-3')
- (Common Sequencing Primers)
Resource Information
-
Articles Citing this Plasmid
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Trademarks:
- Zeocin® is an InvivoGen trademark.
Depositor Comments
Please see the supplemental documents "Plasmid gb file" and "Cloning protocol" for more information on using this plasmid.
To confirm a sgRNA sequence cloned into this plasmid using the BbsI sites, use the hU6-F primer (5'-GAGGGCCTATTTCCCATGATT-3').
These plasmids were created by your colleagues. Please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
-
For your Materials & Methods section:
pU6-(BbsI)_CBh-Cas9-T2A-BFP was a gift from Ralf Kuehn (Addgene plasmid # 64323 ; http://n2t.net/addgene:64323 ; RRID:Addgene_64323) -
For your References section:
Increasing the efficiency of homology-directed repair for CRISPR-Cas9-induced precise gene editing in mammalian cells. Chu VT, Weber T, Wefers B, Wurst W, Sander S, Rajewsky K, Kuhn R. Nat Biotechnol. 2015 Mar 24. doi: 10.1038/nbt.3198. 10.1038/nbt.3198 PubMed 25803306