-
Purpose3rd generation plasmid for phage display on modified geneIII, contains stuffer fragment
-
Depositing Lab
-
Sequence Information
Ordering
| Item | Catalog # | Description | Quantity | Price (USD) | |
|---|---|---|---|---|---|
| Plasmid | 63890 | Standard format: Plasmid sent in bacteria as agar stab | 1 | $94 | |
Backbone
-
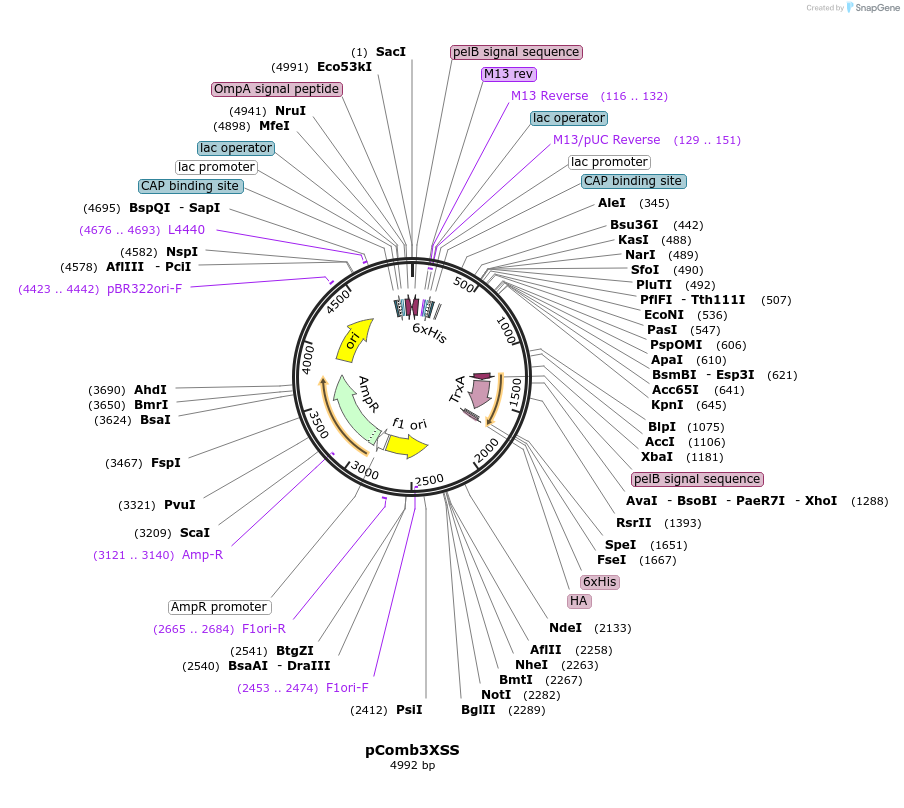
Vector backbonepComb3H
- Backbone size w/o insert (bp) 3300
- Total vector size (bp) 4900
-
Modifications to backboneAddition of 6xHis and HA tags for detection and purification, amber stop codon upstream of geneIII to produce soluble protein by switching to a non-suppressor strain of E. coli (e.g. Top10F')
-
Vector typeBacterial Expression ; phage display
Growth in Bacteria
-
Bacterial Resistance(s)Ampicillin, 100 μg/mL
-
Growth Temperature37°C
-
Growth Strain(s)XL1 Blue
-
Copy numberHigh Copy
Gene/Insert 1
-
Gene/Insert nameSS Stuffer
-
Alt nameSfiI Stuffer
-
SpeciesSynthetic
-
Insert Size (bp)1600
- Promoter lacZ
-
Tags
/ Fusion Proteins
- 6x His (C terminal on backbone)
- HA tag (C terminal on backbone)
- geneIII (C terminal on backbone)
Cloning Information for Gene/Insert 1
- Cloning method Restriction Enzyme
- 5′ cloning site SfiI (not destroyed)
- 3′ cloning site SfiI (not destroyed)
- 5′ sequencing primer 5'-AAG ACA GCT ATC GCG ATT GCA G-3'
- 3′ sequencing primer 5'-GCC CCC TTA TTA GCG TTT GCC ATC-3'
- (Common Sequencing Primers)
Gene/Insert 2
-
Gene/Insert nameLight Chain Stuffer
-
SpeciesSynthetic
-
Insert Size (bp)1200
Cloning Information for Gene/Insert 2
- Cloning method Restriction Enzyme
- 5′ cloning site SacI (not destroyed)
- 3′ cloning site XbaI (not destroyed)
- 5′ sequencing primer 5'-AAG ACA GCT ATC GCG ATT GCA G-3'
- (Common Sequencing Primers)
Gene/Insert 3
-
Gene/Insert nameHeavy Chain Stuffer
-
SpeciesSynthetic
-
Insert Size (bp)300
Cloning Information for Gene/Insert 3
- Cloning method Restriction Enzyme
- 5′ cloning site XhoI (not destroyed)
- 3′ cloning site SpeI (not destroyed)
- 5′ sequencing primer NA
- 3′ sequencing primer 5'-AGA AGC GTA GTC CGG AAC GTC-3'
- (Common Sequencing Primers)
Resource Information
-
Articles Citing this Plasmid
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Trademarks:
- Zeocin® is an InvivoGen trademark.
Depositor Comments
pComb3X is the newest of the pComb vectors. Improvements over pComb3 include increased stability and introduction of an asymmetric SfiI cassette for directional cloning of full Fab, scFv, peptide and other protein for phage display. 6xHis and HA tags allow for purification and detection. An amber stop codon was introduced to turn-off expression of the pIII fusion protein by switching to a non-supressor strain of E. coli allowing production of soluble protein without subcloning. Alternatively, the gene for phage protein pIII can be removed by SpeI/NheI digest. pComb3XSS is recommended for preparation of vector for library cloning. The “SS” refers to the double stuffer, a 1200bp stuffer in the Fab light chain cloning region bounded by SacI and XbaI restriction sites and a 300bp stuffer in Fab heavy chain cloning region bound by XhoI and SpeI restriction sites. Also, the 1600bp double stuffer (both stuffers plus the leader sequence between the Fab light chain and heavy chain cloning regions) can be removed by SfiI digest so that non-Fab genes of interest can be cloned. Also available on Addgene: pComb3XTT and pComb3XLambda are only needed at templates for the construction of chimeric Fab libraries as described in Phage Display: A Laboratory Manual. pComb3XTT can also be used as an Fab expression control.
Select References:
Barbas, C. F., III; Burton, D. R.; Scott, J.K., Silverman, G.J. Eds. (2001) Phage Display: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, New York
Rader, C; Popkov, M.; Neves, J.A.; Barbas III, C.F. (2002) Integrin _v_3 Targeted Therapy of Kaposi’s Sarcoma with an In Vitro Evolved Antibody. FASEB, 16(14):2000-2.
Berry, J.D.; Rutherford, J.; Silverman, G.J.; Kaul, R.; Elia, M.; Gobuty, S.; Fuller, R.; Plummer, F.A.; & Barbas III, C.F. (2003) Development of Functional Human Monoclonal Single-Chain Variable Fragment Antibody Against HIV-1 From Human Cervical B cells. Hybridoma and Hybridomics, 22(2):, 97-108.
Jendreyko N, Popkov M, Beerli RR, Chung J, McGavern DB, Rader C, Barbas CF 3rd. (2003) Intra-diabodies: Bispecific, tetravalent antibodies for the simultaneous functional knockout of two cell surface receptors. J Biol Chem., 278(48):, 47812-9.
Steinberger, P.; Sutton, J.K.; Rader, C.; Elia, M.; and Barbas III, C. F. (2000) Generation and Characterization of a Recombinant Human CCR5-specific Antibody: A Phage Display Approach for Rabbit Antibody Humanization. J. Biol. Chem., 275,, 36073-36078.
Goncalves, J.; Kilva, F.; Freitas-Vieira, A.; Santa-Marta, M.; Malho, R.; Yang, X.; Gabuzda, D.; and Barbas III, C.F. (2002) Functional Neutralization of HIV-1 Vif Protein by Intracellular Immunization Inhibits Reverse Transcription and Viral Replication. J. Biol. Chem., 277(35):32036-45.
Popkov, M.; Mage, R.G.; Alexander, C.B.; Thundivalappil, S.; Barbas III, C.F.; Rader, C. (2003) Rabbit immune repertoires as sources for therapeutic monoclonal antibodies: The impact of Kappa Allotype-correlated variation in cysteine content on antibody libraries selected by phage display. J. Mol. Biol., 325:325-335.
Chung, J.; Rader, C.; Popkov, M.; Hur, Y.-M.; Kim, H.-K.; Lee, Y.-J.; & Barbas III, C. F. (2004) Integrin _IIb_3 specific synthetic human monoclonal antibodies and HCDR3 peptides that potently inhibit platelet aggregation,FASEB 18(2):361-3.
Popkov, M.; Jendreyko, N.; Gonzalez-Sapienza, G.; Mage, R.G.; Rader, C.; Barbas III, C.F. (2004) Human/mouse cross-reactive anti-VEGF receptor 2 recombinant antibodies selected from animmune b9 allotype rabbit antibody library, J. Immunol. Methods, 288(1-2):149-164.
Popkov, M.; Rader, C.; Barbas III, C.F. (2004) Isolation of human prostate cancer reactive
antibodies using phage display technology, J. Immunol. Methods, 291:137-151.
Tanaka, F.; Fuller, R.; Barbas III, C.F. (2005) Development of Small Designer Aldolase Enzymes: Catalytic Activity, Folding, and Substrate Specificity. Biochemistry, 44:7583-7592.
This plasmid has been found to be prone to multimerization. Multimerization often does not impact plasmid function, but may reduce transformation efficiencies. See our article on Plasmid Dimers and Multimers for more information (https://blog.addgene.org/plasmids-101-dimers-and-multimers)
These plasmids were created by your colleagues. Please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
-
For your Materials & Methods section:
pComb3XSS was a gift from Carlos Barbas (Addgene plasmid # 63890 ; http://n2t.net/addgene:63890 ; RRID:Addgene_63890) -
For your References section:
Methods for the generation of chicken monoclonal antibody fragments by phage display. Andris-Widhopf J, Rader C, Steinberger P, Fuller R, Barbas CF 3rd. J Immunol Methods. 2000 Aug 28;242(1-2):159-81. 10.1016/s0022-1759(00)00221-0 PubMed 10986398