-
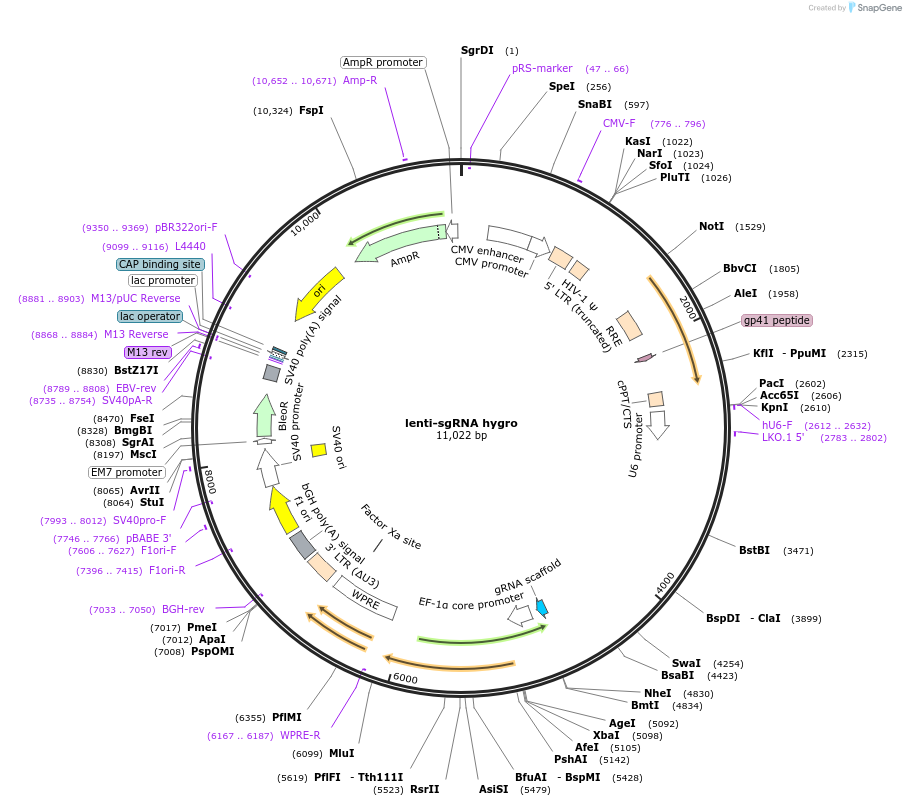
Purpose(Empty Backbone) This 3rd generation lentiviral plasmid expresses a S. pyogenes CRISPR chimeric RNA element with customizable sgRNA from a U6 promoter and hygromycin resistance from an EF-1a promoter.
-
Depositing Lab
-
Sequence Information
Ordering
| Item | Catalog # | Description | Quantity | Price (USD) | |
|---|---|---|---|---|---|
| Plasmid | 104991 | Standard format: Plasmid sent in bacteria as agar stab | 1 | $94 | |
Backbone
-
Vector backbonelentiCRISPRv2 hygro (Addgene #98291)
-
Backbone manufacturerBrett Stringer from Feng Zhang plasmid lentiCRISPR v2 (Addgene plasmid #52961)
- Backbone size (bp) 11022
-
Modifications to backboneThe first of the two MluI restriction sites of lentiCRISPR v2 (Addgene plasmid #52961) was mutated (to create lentiCRISPRv2 puro, Addgene plasmid #98290) to allow the subcloning of alternative P2A-antibiotic resistance cassettes between the BamHI and remaining MluI restriction site (see lentiCRISPRv2 hygro, Addgene plasmid #98291; lentiCRISPRv2 neo, Addgene plasmid #98292; and lentiCRISPRv2 blast, Addgene plasmid #98293). The Cas9-P2A-hygro cassette of lentiCRISPRv2 hygro (Addgene plasmid #98291) was removed by XbaI/MluI digestion and the hygromycin resistance gene sequence was cloned in its place between the XbaI and MluI restriction sites downstream of the EF-1alpha core promoter (see also lentiGuide puro, Addgene plasmid #104990; lentiGuide neo, Addgene plasmid #104992; and lentiGuide blast, Addgene plasmid #104993).
-
Vector typeMammalian Expression, Lentiviral, CRISPR
-
Selectable markersHygromycin
Growth in Bacteria
-
Bacterial Resistance(s)Ampicillin, 100 μg/mL
-
Growth Temperature37°C
-
Growth Strain(s)Stbl4
-
Growth instructionsSingle colonies of transformed STBL3 or STBL4 cells cultured in 5 ml of LB broth containing 100 micrograms/ml ampicillin for 20 hours at 37C, 200 rpm give good yields for plasmid minipreps.
-
Copy numberHigh Copy
Resource Information
-
Addgene Notes
-
A portion of this plasmid was derived from a plasmid made byThe hygromycin resistance gene sequence was amplified by PCR from pF_GVP16_hygro (a gift from John Silke, Cell Death and Differentiation (2008) 15, 213–219). The BsmBI restriction site it contains was removed by site-directed mutagenesis.
-
Articles Citing this Plasmid
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Trademarks:
- Zeocin® is an InvivoGen trademark.
Depositor Comments
This 3rd generation lentiviral plasmid expresses a S. pyogenes CRISPR chimeric RNA element with a customizable sgRNA from a U6 promoter and hygromycin resistance gene from an EF-1a core promoter.
This plasmid does NOT contain Cas9. It should be used in conjunction with lentiCas9 puro, lentiCas9 neo or lentiCas9 blast or with cell lines expressing Cas9.
After cloning a double-stranded oligonucleotide specifying a gRNA between the two BsmBI restriction sites, use hU6-F (5'-GAGGGCCTATTTCCCATGATT-3') or LKO.1 5’(5'- GACTATCATATGCTTACCGT-3') to sequence the chimeric RNA element.
Instruction for gRNA double-stranded oligonucleotide design and cloning can be found in the Zhang lab protocol, https://media.addgene.org/data/plasmids/52/52961/52961-attachment_B3xTwla0bkYD.pdf
These plasmids were created by your colleagues. Please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
-
For your Materials & Methods section:
lenti-sgRNA hygro was a gift from Brett Stringer (Addgene plasmid # 104991 ; http://n2t.net/addgene:104991 ; RRID:Addgene_104991) -
For your References section:
A reference collection of patient-derived cell line and xenograft models of proneural, classical and mesenchymal glioblastoma. Stringer BW, Day BW, D'Souza RCJ, Jamieson PR, Ensbey KS, Bruce ZC, Lim YC, Goasdoue K, Offenhauser C, Akgul S, Allan S, Robertson T, Lucas P, Tollesson G, Campbell S, Winter C, Do H, Dobrovic A, Inglis PL, Jeffree RL, Johns TG, Boyd AW. Sci Rep. 2019 Mar 20;9(1):4902. doi: 10.1038/s41598-019-41277-z. 10.1038/s41598-019-41277-z PubMed 30894629