-
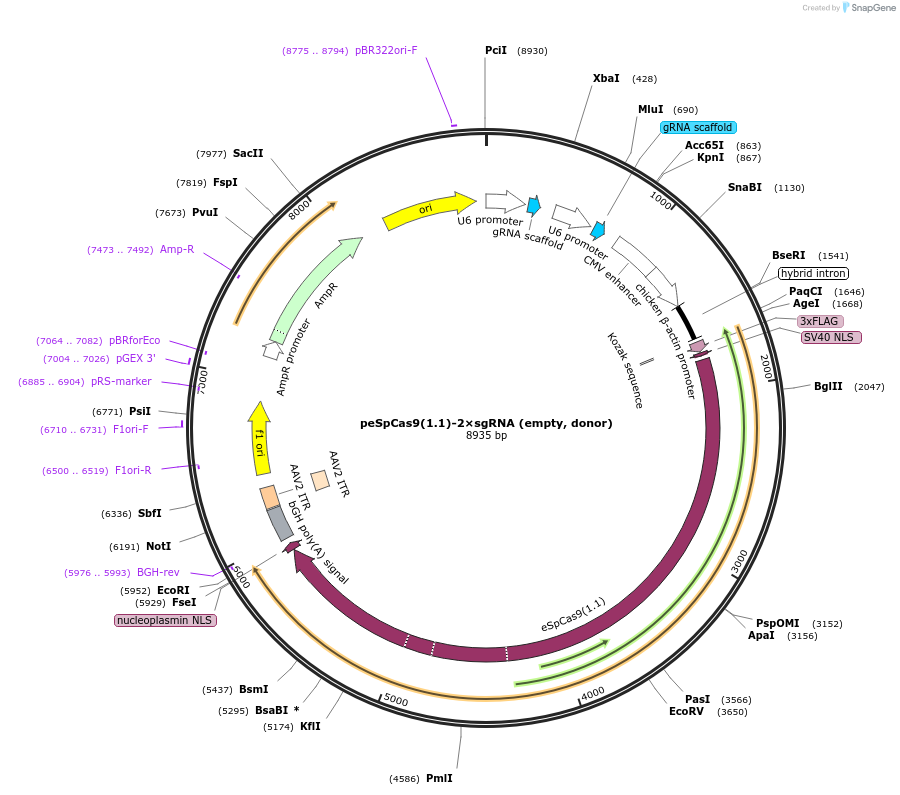
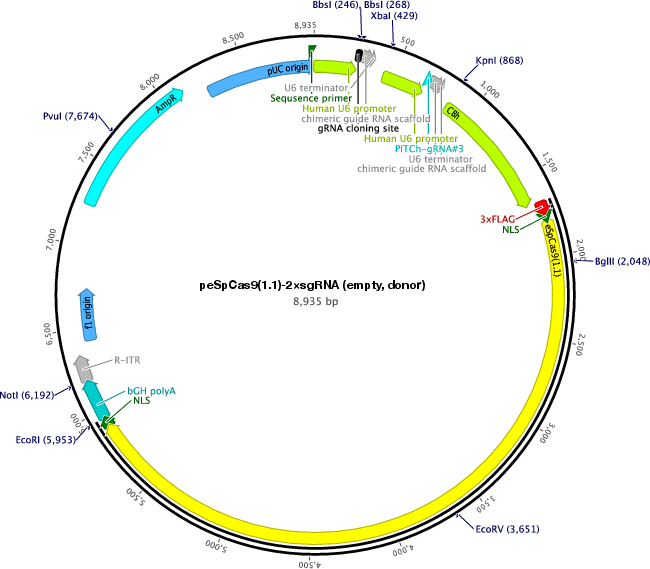
Purpose(Empty Backbone) All-in-one vector for CRISPR/Cas9-mediated homology-independent knock-in system. The plasmid contains eSpCas9(1.1) and two sgRNA expression cassettes. The first gRNA cloning site is empty.
-
Depositing Lab
-
Sequence Information
Ordering
| Item | Catalog # | Description | Quantity | Price (USD) | |
|---|---|---|---|---|---|
| Plasmid | 80768 | Standard format: Plasmid sent in bacteria as agar stab | 1 | $94 | |
Backbone
-
Vector backboneeSpCas9(1.1)
-
Backbone manufacturerFeng Zhang lab (Plasmid #71814)
- Backbone size (bp) 8506
-
Modifications to backboneA second sgRNA expression cassette was added after the first sgRNA cassette in the plasmid.
-
Vector typeMammalian Expression, CRISPR
Growth in Bacteria
-
Bacterial Resistance(s)Ampicillin, 100 μg/mL
-
Growth Temperature37°C
-
Growth Strain(s)DH5alpha
-
Copy numberHigh Copy
Resource Information
-
Articles Citing this Plasmid
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Trademarks:
- Zeocin® is an InvivoGen trademark.
These plasmids were created by your colleagues. Please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
-
For your Materials & Methods section:
peSpCas9(1.1)-2×sgRNA (empty, donor) was a gift from Kazuhisa Nakayama (Addgene plasmid # 80768 ; http://n2t.net/addgene:80768 ; RRID:Addgene_80768) -
For your References section:
Practical method for targeted disruption of cilia-related genes by using CRISPR/Cas9-mediated homology-independent knock-in system. Katoh Y, Michisaka S, Nozaki S, Funabashi T, Hirano T, Takei R, Nakayama K. Mol Biol Cell. 2017 Feb 8. pii: mbc.E17-01-0051. doi: 10.1091/mbc.E17-01-0051. 10.1091/mbc.E17-01-0051 PubMed 28179459