-
Purpose(Empty Backbone)
-
Depositing Lab
-
Publication
-
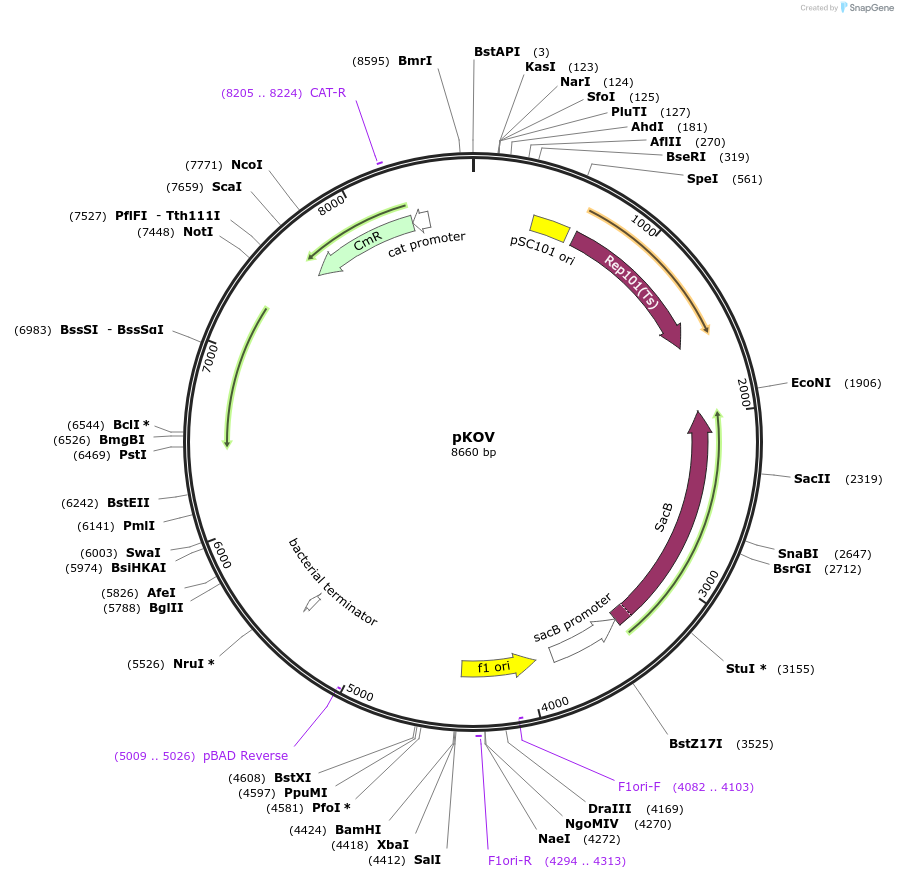
Sequence Information
Ordering
| Item | Catalog # | Description | Quantity | Price (USD) | |
|---|---|---|---|---|---|
| Plasmid | 25769 | Standard format: Plasmid sent in bacteria as agar stab | 1 | $94 | |
Backbone
-
Vector backbonepMAK700, pMAK705, pBS-TS
-
Backbone manufacturerN/A
- Backbone size (bp) 8673
-
Vector typeBacterial Expression
Growth in Bacteria
-
Bacterial Resistance(s)Chloramphenicol, 25 μg/mL
-
Growth Temperature30°C
-
Growth Strain(s)Top10
-
Growth instructionsGrow at 30 degrees Celsius
-
Copy numberUnknown
Gene/Insert
-
Gene/Insert nameNone
-
Alt namepKOV gene replacement vector
-
Alt namepKO5
Cloning Information
- Cloning method Restriction Enzyme
- 5′ sequencing primer pK03-L: 5'-AGGGCAGGGTCGTTAAATAGC-3'
- 3′ sequencing primer pK03-R: 5'-TTAATGCGCCGCTACAGGGCG-3'
- (Common Sequencing Primers)
Resource Information
-
Reference
-
Articles Citing this Plasmid
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Trademarks:
- Zeocin® is an InvivoGen trademark.
Depositor Comments
pKOV is identical to the pKO3 vector described in J. Bacteriology 179: 6228-6237, except for the addition of a 3kb stuffer sequence in the multiple cloning site. This stuffer permits (i) directional cloning using NotI and BamHI and (ii) clean separation of doubly cut vector from singly cut contaminants when using this pair of enzymes. The pKOV cloning site is: 5' - SmaI - NotI - SmaI- stuffer - BamHI - SalI - 3'. BamHI and SalI are not used together, nor is SmaI used with NotI. BglII & BclI cut ends are compatible with BamHI. PmeI & SwaI are compatible with SmaI.
NOTE: pKOV has a temperature sensitive pSC101 replication origin. To recover the plasmid, strains harboring the plasmid must be grown at 30 deg C under chloramphenicol selection.
Gene replacement: Mutant alleles cloned into the pKOV gene replacement vector are electroporated into recombination proficient strains (eg. EMG2) and allowed to recover for 1 h at 30 deg C. The cells are plated on prewarmed chloramphenicol/LB plates and incubated at 42 deg C. To measure the integration frequency, the electroporated cells are also plated on chloramphenicol/LB plates at 30 deg C. From the 42 deg C plate, 1-5 colonies are picked into 1 ml of LB broth, serially diluted, and immediately plated at 30°ree;C on either 5% w/v sucrose or 5% sucrose+antibiotic plates. The 5% sucrose plates are replica plated to chloramphenicol plates at 30 deg C to test for loss of the replacement vector (cms). The gene replacement is confirmed by either PCR using primers flanking the targeted open reading frame or by genomic Southern's.
Note from depositor: If sucrose plates do not select correctly, pKOV should be used in liquid selection on a plate reader with a LB + Cm control growth and a LB + Cm + sucrose growth. pKOV can sometimes still allow survival in the presence of sucrose, but significantly decreases fitness, so tracking the kinetic growth can be helpful.
The sacB gene loses its efficacy,, sacB is toxic in E. coli even in the absence of sucrose.
These plasmids were created by your colleagues. Please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
-
For your Materials & Methods section:
pKOV was a gift from George Church (Addgene plasmid # 25769 ; http://n2t.net/addgene:25769 ; RRID:Addgene_25769) -
For your References section:
Methods for generating precise deletions and insertions in the genome of wild-type Escherichia coli: application to open reading frame characterization. Link AJ, Phillips D, Church GM. J Bacteriol. 1997 Oct . 179(20):6228-37. PubMed 9335267