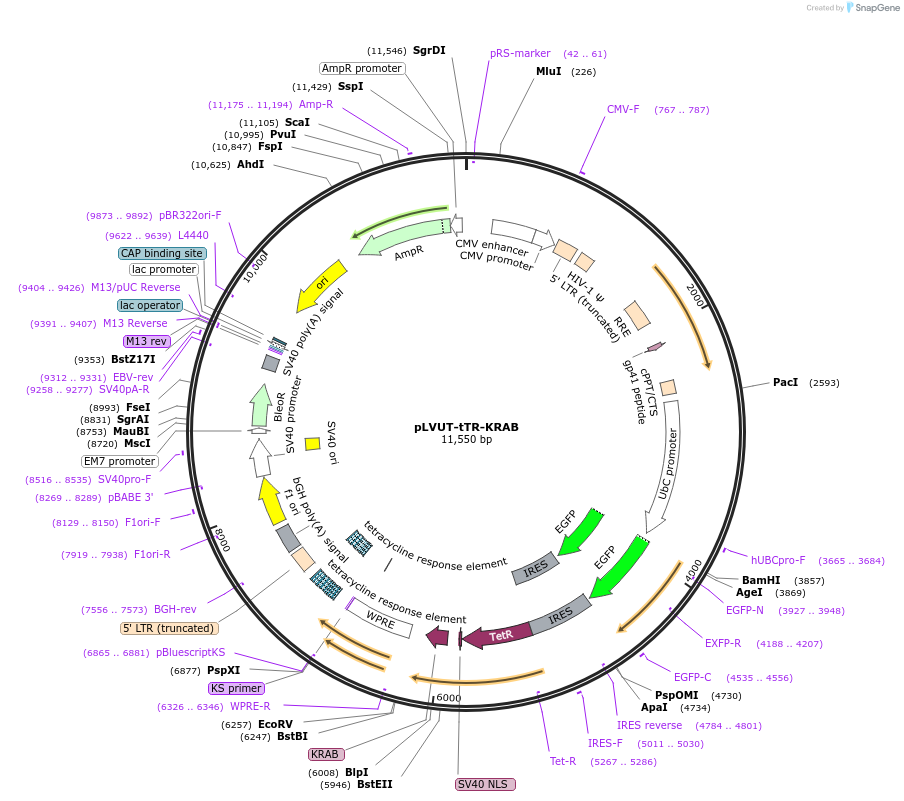
-
PurposeTet-regulated (Tet-on) lentiviral vector for transgene (hUbiquitin promoter) - AND/OR - shRNA (H1 promoter when subcloned from pLVTHM (Addgene#12247)) - 3rd generation
-
Depositing Labs
-
Sequence Information
Ordering
| Item | Catalog # | Description | Quantity | Price (USD) | |
|---|---|---|---|---|---|
| Plasmid | 11651 | Standard format: Plasmid sent in bacteria as agar stab | 1 | $94 | |
| Cloning Grade DNA | 11651-DNA.cg | 2 µg of cloning grade DNA in Tris buffer | 1 | $115 | |
Backbone
-
Vector backboneNone
- Backbone size w/o insert (bp) 11480
-
Vector typeMammalian Expression, Lentiviral
Growth in Bacteria
-
Bacterial Resistance(s)Ampicillin, 100 μg/mL
-
Growth Temperature37°C
-
Growth Strain(s)Stbl3
-
Growth instructionsUse Stbl3 or HB101 to reduce chance of recombination. Grow at 37C
-
Copy numberHigh Copy
Gene/Insert
-
Gene/Insert namehUbiquitin C, GFP, tTR-KRAB, Tet-on
Cloning Information
- Cloning method Restriction Enzyme
- 5′ sequencing primer See map
- (Common Sequencing Primers)
Resource Information
-
Articles Citing this Plasmid
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Trademarks:
- Zeocin® is an InvivoGen trademark.
Depositor Comments
Transgenes can be expressed from any RNA Pol II promoter as part of bicistronic unit comprising the KRAB-based repressor; tetO sequences are inserted into the vector LTR. Tet-on and Tet-off versions rely on repressors that bind in the absence or the presence of doxycycline, respectively. Addition of Pol III promoter-small hairpin RNA cassette allows for drug-controllable RNA interference (Tet-on shRNA).
pLVTHM and packaging plasmid for this system are also available at Addgene http://www.addgene.org/rnaitools Please visit Trono lab website http://tronolab.epfl.ch to see frequently asked
questions on cloning strategies and packaging.
Note: There is another EcoRI site at 6235 that is not depicted in the author's map.
Cloning Grade DNA (Catalog # 11651-DNA.cg)
Purpose
Cloning grade DNA is suitable for use in PCR, cloning reactions, or transformation into E. coli. The purity and amount is not suitable for direct transfections.
Delivery
- Amount 2 µg
- Guaranteed Concentration 100 ng/µl +/- 5 ng/µl
- Pricing $115 USD
- Storage DNA can be stored at 4℃ (short term) or -20℃ (long term).
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Quality Control
Addgene has verified this plasmid using Next Generation Sequencing. Results are available here
These plasmids were created by your colleagues. Please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
-
For your Materials & Methods section:
pLVUT-tTR-KRAB was a gift from Patrick Aebischer & Didier Trono (Addgene plasmid # 11651 ; http://n2t.net/addgene:11651 ; RRID:Addgene_11651) -
For your References section:
A versatile tool for conditional gene expression and knockdown. Szulc J, Wiznerowicz M, Sauvain MO, Trono D, Aebischer P. Nat Methods. 2006 Feb;3(2):109-16. doi: 10.1038/nmeth846 10.1038/nmeth846 PubMed 16432520