 p53 Pathway
p53 Pathway
Background
p53 is a transcription factor and tumor suppressor activated by cellular stresses like DNA damage, oxidative stress, and nutrient deprivation. p53 is activated through both phosphorylation and acetylation, which disrupt its interaction with negative regulators, increase its stability and DNA binding activity, and allow it to bind transcriptional co-activators and modulate transcription. The list of p53 transcriptional targets is ever-expanding; the best-characterized effects of p53 are in promoting cell cycle arrest, apoptosis, or senescence in damaged cells. The p53 name arose from its observed molecular weight (53 kDa) when run on an SDS-PAGE gel.
Cancer Impact
p53 is the most frequently mutated gene (>50%) in human cancer; most of these mutations are missense mutations in the DNA-binding domain, often at one of six hotspot residues. These mutations can be inherited or arise through exposure to mutagens like radiation or viruses (e.g., HPV, the human papillomavirus). These mutations interfere with p53’s ability to activate transcription, and they also have a dominant negative effect on functional p53 through oligomerization. In particular, the loss of p53’s pro-apoptotic effects is especially important to tumorigenesis. Inheriting only one functional copy of the p53 gene causes a large reduction in tumor suppression activity, leading to the development of tumors in early adulthood, a disorder known as Li-Fraumeni syndrome. Recently some impacts of p53 mutations have been characterized as gain-of-function, expanding the mechanisms by which mutant p53 is known to function in cancer.
p53 Pathway Plasmids
Click on a name to find available plasmids for the gene, or browse the gene list below. For multiple isoforms or subunits, individual links to each gene page are provided below. Color is used for clarity and does not indicate a specific relationship.
p53 Pathway
Color is used for clarity and does not indicate a specific relationship.
The content for this page was generated with the help of .
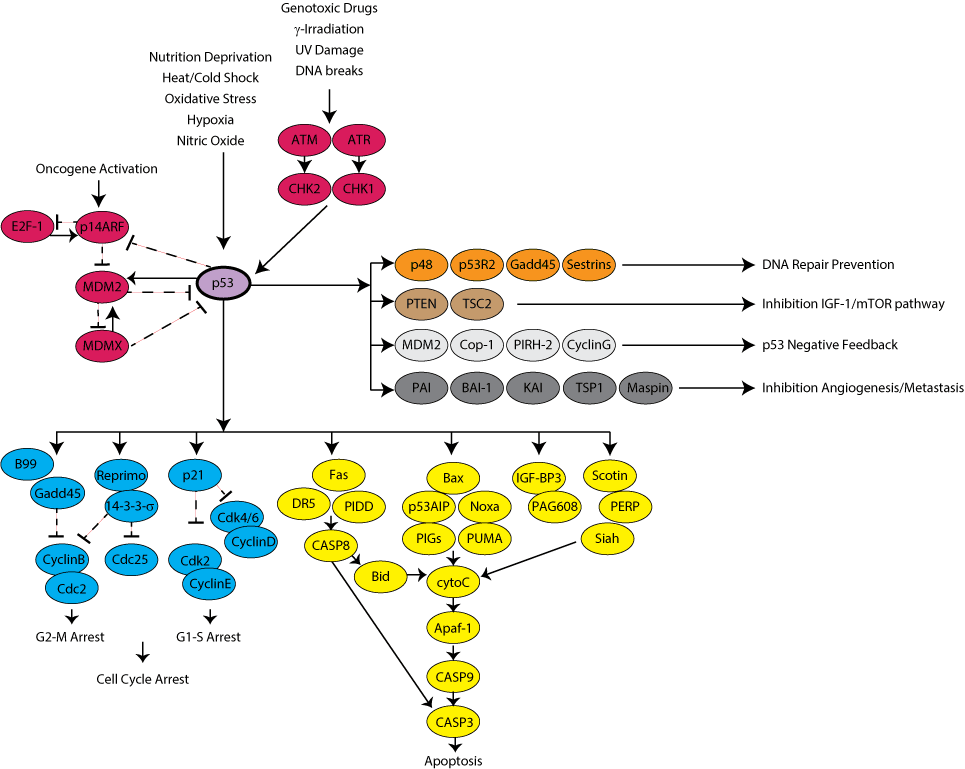
p53 Pathway - Gene List
Click on a name to find available plasmids for the gene. For components with multiple isoforms or subunits, individual links to each gene page are provided below.
| Symbol | Name |
|---|---|
| 14-3-3-σ | Stratifin |
| Apaf-1 | Apoptotic peptidase activating factor 1 |
| ATM | ATM serine/threonine kinase |
| ATR | ATR serine/threonine kinase |
| B99 | G-2 and S-phase expressed 1; also known as GTSE1 |
| BAI-1 | Brain-specific angiogenesis inhibitor 1 |
| Bax | BCL2-associated X protein |
| Bid | BH3 interacting domain death agonist |
| CASP3 | Caspase 3, apoptosis-related cysteine peptidase |
| CASP8 | Caspase 8, apoptosis-related cysteine peptidase |
| CASP9 | Caspase 9, apoptosis-related cysteine peptidase |
| Cyclin B | Cyclin B1, 2, or 3 |
| Cyclin D | Cyclin D1, 2, or 3 |
| Cyclin E | Cyclin E1 or E2 |
| Cyclin G | Cyclin G1 or G2 |
| CDC25 | Cell division cycle 25C |
| Cdk4/6 | Cyclin-dependent kinase 4 or 6 |
| CHK1 | Checkpoint kinase 1 |
| CHK2 | Checkpoint kinase 2 |
| Cop-1 | Ring finger and WD repeat domain 2 (RFWD2); E3 ubiquitin protein ligase |
| cytoC | Cytochrome c |
| DR5 | Tumor necrosis factor receptor superfamily, member 10b |
| E2F-1 | E2F transcription factor 1 |
| Fas | Fas cell surface death receptor |
| Gadd45 | Growth arrest and DNA-damage-inducible; alpha, beta, or gamma |
| IGF-BP3 | Insulin-like growth factor binding protein 3 |
| KAI | CD82 molecule |
| Maspin | Serpin family B member 5 |
| MDM2 | MDM2 proto-oncogene, E3 ubiquitin protein ligase |
| MDMX | p53 regulator; also known as MDM4 |
| Noxa | Phorbol-12-myristate-13-acetate-induced protein 1; also known as PMAIP1 |
| p14ARF | Cyclin-dependent kinase inhibitor 2A |
| p21 | Cyclin-dependent kinase inhibitor 1A |
| p48 | Damage-specific DNA binding protein 2, 48kDa |
| p53 | Tumor protein p53 |
| p53AIP1 | Tumor protein p53 regulated apoptosis inducing protein 1 |
| p53R2 | p53 inducible, ribonucleotide reductase M2 B (RRM2B) |
| PAG608 | Zinc finger, matrin-type 3 |
| PAI | Serpin peptidase inhibitor, clade E (nexin, plasminogen activator inhibitor type 1), member 1 |
| PERP | TP53 apoptosis effector |
| PIDD | p53-induced death domain protein 1 |
| PIGs | Etoposide induced 2.4 |
| PIRH-2 | Ring finger and CHY zinc finger domain containing 1 (RCHY1); E3 ubiquitin protein ligase |
| PTEN | Phosphatase and tensin homolog |
| PUMA | BCL2 binding component 3 |
| Reprimo | TP53 dependent G2 arrest mediator candidate |
| Scotin | Shisa family member 5 |
| Sestrins | Sestrins 1, 2, or 3 |
| Siah | Siah E3 ubiquitin protein ligase 1 |
| TSC2 | Tuberous sclerosis 2 |
| TSP1 | Thrombospondin 1 |
References
Unravelling mechanisms of p53-mediated tumour suppression. Bieging KT, Mello SS, Attardi LD. Nat Rev Cancer. 2014 May;14(5):359-70. PubMed PMID: 24739573.
Uncovering the role of p53 splice variants in human malignancy: a clinical perspective. Surget S, Khoury MP, Bourdon JC. Onco Targets Ther. 2013 Dec 19;7:57-68. doi: 10.2147/OTT.S53876. PubMed PMID: 24379683.
When mutants gain new powers: news from the mutant p53 field. Brosh R, Rotter V. Nat Rev Cancer. 2009 Oct;9(10):701-13. PubMed PMID: 19693097 .
The expanding universe of p53 targets. Menendez D, Inga A, Resnick MA. Nat Rev Cancer. 2009 Oct;9(10):724-37. PubMed PMID: 19776742.
Germline TP53 mutations and Li-Fraumeni syndrome. Varley JM. Hum Mutat. 2003 Mar;21(3):313-20. PubMed PMID: 12619118.
p53 mutant mice that display early ageing-associated phenotypes. Tyner SD, Venkatachalam S, Choi J, Jones S, Ghebranious N, Igelmann H, Lu X, Soron G, Cooper B, Brayton C, Park SH, Thompson T, Karsenty G, Bradley A, Donehower LA. Nature. 2002 Jan 3;415(6867):45-53. PubMed PMID: 11780111.
p53 mutations in human cancers. Hollstein M, Sidransky D, Vogelstein B, Harris CC. Science. 1991 Jul 5;253(5015):49-53. PubMed PMID: 1905840.
Recruitment of p300/CBP in p53-dependent signal pathways. Avantaggiati ML, Ogryzko V, Gardner K, Giordano A, Levine AS, Kelly K. Nature. 1997 Jun 27;89(7):1175-84. PubMed PMID: 9215639.
Mutant p53 gain-of-function in cancer. Oren, M. and V. Rotter. Cold Spring Harbor Perspectives in Biology. 2010 Feb;2(2):a001107. PMC PMID: 20182618.
Do you have suggestions for other plasmids that should be added to this list?
Fill out our Suggest a Plasmid form or e-mail [email protected] to help us improve this resource!



