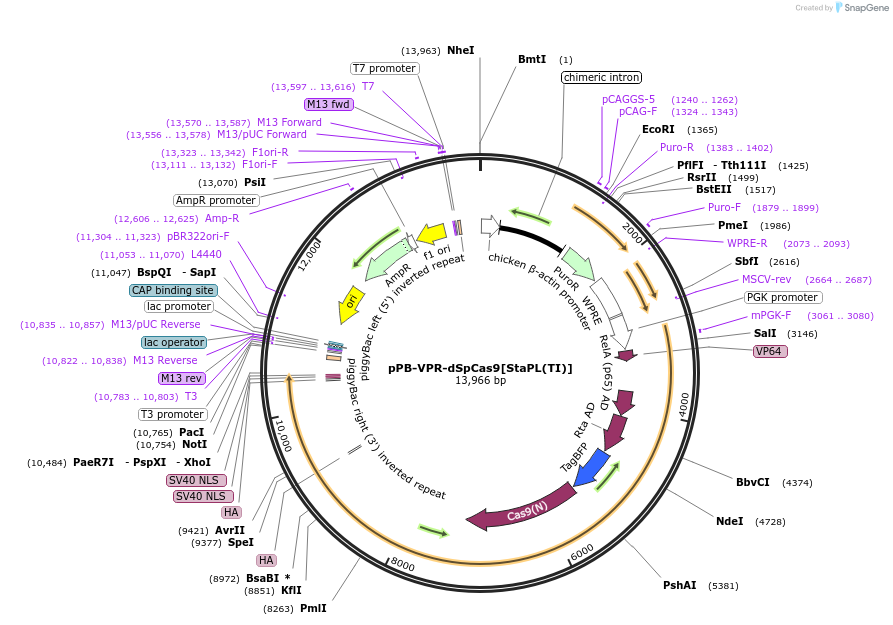
pPB-VPR-dSpCas9[StaPL(TI)]
(Plasmid
#111508)
-
PurposeExpresses a VPR transcriptional activator fused to the nuclease-deactivated S. pyogenes Cas9, which is governed by an inserted StaPL(TI) module, in mammalian cells. [TI = telaprevir inhibited].
-
Depositing Lab
-
Sequence Information
Ordering
| Item | Catalog # | Description | Quantity | Price (USD) | |
|---|---|---|---|---|---|
| Plasmid | 111508 | Standard format: Plasmid sent in bacteria as agar stab | 1 | $94 | |
Backbone
-
Vector backbonepPB
-
Backbone manufacturerSystem Biosciences
- Total vector size (bp) 13958
-
Vector typeMammalian Expression
-
Selectable markersPuromycin
Growth in Bacteria
-
Bacterial Resistance(s)Ampicillin, 100 μg/mL
-
Growth Temperature37°C
-
Growth Strain(s)DH5alpha
-
Copy numberUnknown
Gene/Insert
-
Gene/Insert nameVPR-dSpCas9[StaPL(TI)]
-
SpeciesH. sapiens (human); Hepatitis C Virus (HCV) genotype 1a, Entacmaea quadricolor, Streptococcus pyogenes, Herpes simplex virus, Epstein-Barr virus
-
Insert Size (bp)7323
-
MutationThe HCV NS3 protease carries F43L, Q80K, and S122R, and D168Y mutations. The VPR domain's internal NLS sequence was mutated to a GGSGGSGGS linker. SpCas9 contains D10A and H840A mutations.
-
GenBank IDNC_004102
- Promoter PGK
Cloning Information
- Cloning method Gibson Cloning
- 5′ sequencing primer CCACCTTCTACTCCTCCC
- 3′ sequencing primer ATTAACCCTCACTAAAGGG
- (Common Sequencing Primers)
Resource Information
-
A portion of this plasmid was derived from a plasmid made bythe Vladislav Verkhusha lab [TagBFP, Ref. 1], the George Church lab [VPR, Ref. 2], and the Stanley Qi lab [dSpCas9, Ref. 3].
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Trademarks:
- Zeocin® is an InvivoGen trademark.
Depositor Comments
[Ref. 1]
SUBACH O.M., GUNDOROV I.S., YOSHIMURA M., SUBACH F.V., ZHANG J., GRUENWALD D., SOUSLOVA E.A., CHUDAKOV D.M., & VERKHUSHA V.V. 2008. Conversion of Red Fluorescent Protein into a Bright Blue Probe. Chem Biol 15 (10):1116-24.
[Ref. 2]
CHAVEZ A., SCHEIMAN J., VORA S., PRUITT B.W., TUTTLE M., P R IYER E., LIN S., KIANI S., GUZMAN C.D., WIEGAND D.J., TER-OVANESYAN D., BRAFF J.L., DAVIDSOHN N., HOUSDEN B.E., PERRIMON N., WEISS R., AACH J., COLLINS J.J. & CHURCH G.M. 2015. Highly efficient Cas9-mediated transcriptional programming. Nat Methods 12: 326-328.
[Ref. 3]
GAO Y., XIONG X., WONG S., CHARLES E.J., LIM W.A. & QI L.S. 2016. Complex transcriptional modulation with orthogonal and inducible dCas9 regulators. Nat Methods 13: 1043-1049.
These plasmids were created by your colleagues. Please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
-
For your Materials & Methods section:
pPB-VPR-dSpCas9[StaPL(TI)] was a gift from Michael Lin (Addgene plasmid # 111508 ; http://n2t.net/addgene:111508 ; RRID:Addgene_111508) -
For your References section:
StaPLs: versatile genetically encoded modules for engineering drug-inducible proteins. Jacobs CL, Badiee RK, Lin MZ. Nat Methods. 2018 Jul;15(7):523-526. doi: 10.1038/s41592-018-0041-z. Epub 2018 Jul 2. 10.1038/s41592-018-0041-z PubMed 29967496