Tetracycline (Tet) Inducible Expression
Inducible and repressible promoters are powerful tools to control gene expression and study biological targets. The tetracycline (tet) system is one of the most popular choices for rapid, efficient, and reversible control of gene expression in eukaryotes.
To avoid secondary effects from repurposing endogenous systems that respond to natural stimuli like hormones or metal ions, scientists have adapted an antibiotic resistance operon from E. coli: the tetracycline repressor protein (TetR) and operator (tetO, a 19 nucleotide sequence, TCCCTATCAGTGATAGAGA). In normal conditions, the TetR protein binds to tetO, blocking transcription of the downstream gene. If tet or one of its analogs like doxycycline (dox) is present, TetR binds to tetracycline and releases tetO, enabling transcription (Figure 1).
While TetR and tetO could be a basic tool to control gene expression, the system has been engineered with a variety of improvements. A synthetic Tet Response Element (TRE) typically features seven copies of tetO upstream of a minimal CMV promoter, and other tet- or dox-dependent promoters (sometimes generally called pTet) have been designed by combining tetO sequences with elements like the H1 RNA promoter. And, most importantly, the TetR protein has been modified to provide two classes of tet-responsive systems: Tet-Off and Tet-On.
Read on to learn more about the components of tet systems and how to use them, or skip ahead to view highlighted Tet plasmids.
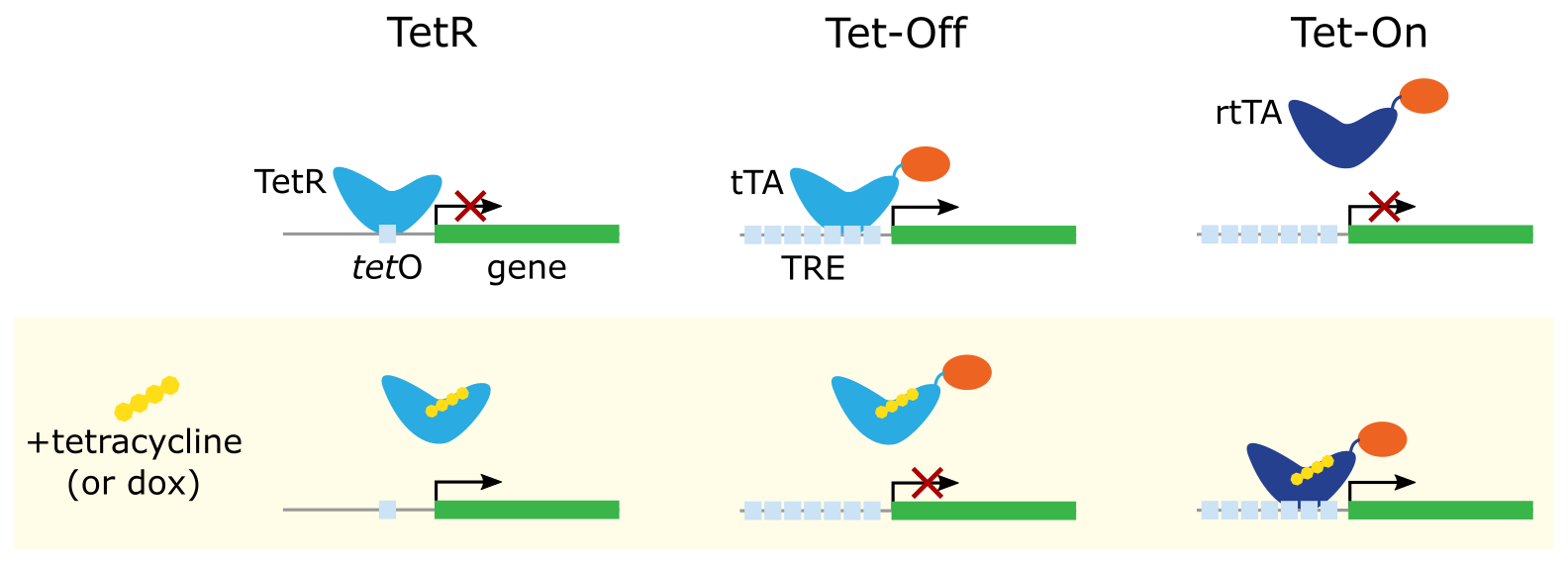
Tetracycline Off (Tet-Off)
The first major advance was the Tet-Off system. A tetracycline-controlled transactivator (tTA) was created by fusing TetR with the activation domain of the herpes simplex virus VP16 protein (Gossen and Bujard, 1992). This work also introduced the TRE, placing seven copies of tetO upstream of the minimal CMV promoter.
In the absence of tetracycline, tTA binds to the TRE and its VP16 domain promotes gene expression. In the presence of tetracycline, tTA unbinds from the TRE, resulting in reduced gene expression. Tetracycline Off is also known as the tTA-dependent or tet-repressible system (or dox-repressible system).
Tetracycline On (Tet-On)
In 1995, Gossen et al. demonstrated that mutating the amino acid residues of TetR that were important for tetracycline-dependent repression led to a reverse Tet repressor, or rTetR, which specifically binds the TRE in the presence of tetracycline. A new reverse tetracycline-controlled transactivator (rtTA) was created by fusing rTetR with VP16.
In the absence of tetracycline, rtTA is unable to bind the TRE. In the presence of tetracycline, rtTA binds the TRE, promoting gene expression. Tetracycline On is also known as the rtTA-dependent or tet-inducible system (or dox-inducible system).
The outcome of Tet-On is similar to the natural TetR-tetO system, even though the DNA-binding profile is reversed (Figure 1). However, rtTA can induce much stronger expression of the downstream gene via its activation domain than simply removing a repressor. Tet-On has since become one of the most popular methods to induce gene expression in experimental systems.
2nd and 3rd Generation Systems
Subsequent efforts have improved the components of these systems for better efficiency, sensitivity, and control of expression. These include:
- Tet-On Advanced: improved transactivator (rtTA-Advanced, also known as rtTA2S-M2) with greater sensitivity to doxycycline and three copies of a minimal activation domain from VP16 for increased induction of gene expression and reduced side effects (Urlinger et al., 2000).
- rtTA3 and Tet-On 3G: further improved rtTA variants with lower background activity and greater sensitivity to doxycycline (Zhou et al., 2006). Tet-On 3G systems can achieve >10,000-fold induction of the gene of interest.
- Tight TRE promoter or PTight: reduced background expression and tighter binding of Tet-Advanced transactivator (developed by Clontech, based on Resnitzky et al., 1994).
- TRE3G promoter: also optimized into PTRE3GV for viral expression and PTRE3GS for all-in-one systems (Loew et al., 2010; Heinz et al., 2011), engineered to eliminate binding of endogenous mammalian transcription factors to further minimize background expression.
- Improved expression of the transactivator proteins in mammalian cells or other species by codon optimization of their sequence and eliminating interactions with endogenous factors.
Experimental Considerations
Expression System
- If your gene of interest should only be expressed occasionally, use Tet-On: express an rtTA or TetR and include a tet-responsive promoter upstream of your gene of interest.
- If your gene of interest should be generally active and only turned off occasionally, use Tet-Off: express a tTA and include a tet-responsive promoter upstream of your gene of interest.
- Many plasmids use "TetO" or "Tet-O" in their name or description, meaning they contain a TRE or tetO and can serve as either Tet-Off or Tet-On systems, depending on whether they are used alongside a tTA or rtTA, respectively.
You should also choose between:
- Dual vector system: express the transactivator and the tet-controlled gene of interest from two separate vectors. Stable cell lines or transgenic animals can be made to achieve greater control over expression with flexibility for your experiments. For example, you could integrate a transactivator under a tissue-specific promoter and administer the transgene separately.
- All-in-one system: express both the transactivator and the tet-controlled gene of interest from one plasmid. A single vector approach may not have as much flexibility and control as a dual vector design, but it is easier to implement and ensures that all transfected or transduced cells express both components in the same relative ratio.
Different generations of transactivators and promoters are generally cross-compatible. Your choice of transactivator, promoter, and type of transduction (transient or stable) may depend on the desired sensitivity, level of induction, or amount of background expression. For example, only a relatively low level of doxycycline can access the brain so a highly sensitive transactivator may be needed. Or, when inducing a potentially toxic protein you may need extremely minimal background expression and only moderate induction. See Das et al., 2016 (Link opens in a new window) for comparison of Tet-On systems in different applications.
Use of Tetracycline or a Derivative
Doxycycline, a derivative of tetracycline, is a preferred effector for tet-responsive systems. Dox binds with high affinity to TetR and derivatives like rTetR, rtTA, and tTA. Dox has good tissue distribution, low toxicity, a known half-life (24 hours), and is relatively inexpensive. The induction profiles for dox and other tetracycline derivatives can be found in Krueger et al., 2004. Some cell culture sera may contain tetracycline or its derivatives which can result in unintended induction. Check the manufacturer’s details to know whether your serum is tetracycline-free.
Highlighted Plasmids
Browse the tables below to find popular empty backbones, expression vectors for transactivators, and vectors for other applications, or search our collection for all dox-regulated plasmids.
Looking for viral preps? Find in-stock viral preps of tetracycline transactivators and inducible tools or consider our AAV Packaged on Request service available for many transactivator or tet-inducible vectors.
Tetracycline-inducible Backbones
Find backbones or constructs to use for cloning to control expression of your gene of interest.
| ID | Plasmid | Description | Transactivator | Promoter | PI | |
|---|---|---|---|---|---|---|
| 21915 | Tet-pLKO-puro | Lentiviral Tet-On plasmid for inducible expression of shRNA with puromycin selection. See Plasmid #21916 for neomycin selection. | TetR | H1-2O2 | Dmitri Wiederschain | |
| 85966 | EZ-Tet-pLKO-Puro | Lentiviral Tet-On plasmid for easier cloning and design of inducible shRNA expression, with puromycin selection. See Plasmid #85973 for blasticidin and Plasmid #85972 for hygromycin selection. | TetR | H1-2O2 | Cindy Miranti | |
| 104321 | tet-pLKO-sgRNA-puro | Lentiviral Tet-On plasmid for inducible expression of sgRNA | TetR | H1-2O2 | Nathanael Gray | |
| 35625 | pAAV-Ptet-RFP-shR-rtTA | AAV Tet-On shRNA vector. To evaluate shRNA efficacy by fluorescence, use Plasmid #35626 as a reporter for cDNA target. | rtTA3 | Ptet, miniCMV | Howard Gu | |
| 44012 | pInducer20 | Lentiviral Tet-On vector for inducible expression. See article (Meerbrey et al., 2011) for additional tookit plasmids for shRNA or gene expression. | rtTA-Advanced | H1-2O2 | Stephen Elledge | |
| 111177 | LT3GEPIR | Lentiviral Tet-On inducible RNAi with miR-E (miR-30 variant)-based backbone | rtTA-Advanced | TRE3G | Johannes Zuber | |
| 60495 | pSBtet-GP | Sleeping Beauty transposon system with luciferase in cloning site. See article (Kowarz et al., 2015) for additional selection markers. | rtTA-Advanced | Tight TRE | Eric Kowarz | |
| 96930 | XLone-GFP | Tet-On PiggyBac vector for inducble expression of EGFP | Tet-On 3G rtTA | TRE3GS | Xiaojun Lian | |
| 171123 | pLVX-TetOne-Puro-GFP | Lentiviral Tet-On vector for inducible expression of EGFP | Tet-On 3G rtTA | TRE3GS | Jason Sheltzer | |
| 11651 | pLVUT-tTR-KRAB | Lentiviral Tet-On vector for inducible expression of transgene and/or shRNA. See article (Szulc et al., 2006) for Tet-Off and additional plasmids. | tetR-KRAB | TRE | Patrick Aebischer, Didier Trono | |
| 41393 | pCW57.1 | Lentiviral Tet-On vector for inducible expression; insert with Gateway cloning and puromycin selection | rtTA-Advanced | Tight TRE | David Root | |
| 100521 | pCW57.1-MAT2A | Lentiviral Tet-Off vector for repressible expression with blasticidin selection. *Note: this vector contains an insert (MAT2A) which can be replaced by another gene of interest. | tTA-Advanced | Tight TRE | David Sabatini | |
| 19407 | pTREtight2 | Empty backbone for Tet-controlled expression (On or Off) | None | Tight TRE | Markus Ralser | |
| 64238 | pTet-IRES-EGFP | Lentiviral plasmid for Tet-controlled expression of transgene of interest with EGFP (On or Off) | None | TRE, miniCMV | Maria Lung | |
| 16542 | pBI-MCS-EGFP | Bidirectional promoter (Pbi) for Tet-responsive expression (On or Off) of both your gene of interest and EGFP. Pbi contains a TRE between two minimal CMV promoters. | None | Pbi (TRE, miniCMV) | Bert Vogelstein |
Transactivators (tTA or rtTA)
Find a construct that expresses the transactivator for your tetracycline system.
| ID | Plasmid | Description | Transactivator | PI | |
|---|---|---|---|---|---|
| 26429 | pLenti CMV rtTA3 Blast (w756-1) | Lentiviral Tet-On vector with blasticidin selection. See Plasmid #26730 for hygromycin selection. | rtTA3 | Eric Campeau | |
| 128061 | pLVX-Tet3G blasticidin | Lentiviral Tet-On vector with CMV promoter | Tet-On 3G rtTA | Oskar Laur | |
| 96963 | pCAG-TetON-3G | Mammalian expression of the Tet-On 3G transactivator | Tet-On 3G rtTA | Elena Cattaneo | |
| 104543 | PB-EF1a-TetOn3G | PiggyBac vector expressing Tet-On 3G transactivator constitutively expressed by EF1a promoter | Tet-On 3G rtTA | David Vereide | |
| 120309 | pAAV-FAH-rtTA3G | AAV vector to express Tet-On 3G transactivator under the c-Fos promoter | Tet-On 3G rtTA | Bong-Kiun Kaang | |
| 20342 | FUW-M2rtTA | Lentiviral Tet-On vector, expressing rtTA from UbC promoter | rtTA-Advanced | Rudolf Jaenisch | |
| 25434 | pMA2640 | Retroviral Tet-On vector for CMV-driven rtTA with EGFP and Blasticidin selection | rtTA-Advanced | Mikhail Alexeyev | |
| 17492 | pLenti CMV TetR Blast (716-1) | Lentiviral Tet-On vector expressing TetR from CMV promoter | TetR | Eric Campeau, Paul Kaufman | |
| 175274 | pAAV-rtTA | AAV Tet-On vector with neuron-specific expression of rtTA3 from hSyn promoter | rtTA3 | Wei Xu | |
| 26803 | pEnt L1L3 EF1a-tTA-2 | Gateway entry vector to express tTA from EF1α promoter, for Tet-Off. See Plasmid #27106 for rtTA. | tTA | Edward Hsiao | |
| 99118 | pAAV-CAG-tTA | AAV Tet-Off vector, expresses tTA from the CAG promoter. See article (Chan et al., 2017) for other viral tTA plasmids. | tTA | Viviana Gradinaru | |
| 104102 | pCAG-tTA | Mammalian expression of tTA from the CAG promoter for Tet-Off | tTA-Advanced | Takeshi Imai | |
| 104109 | pAAV-Syn1-tTA | AAV Tet-Off vector, expressed tTA from the hSyn promoter | tTA-Advanced | Takeshi Imai |
Other Tet Applications
Explore some other popular applications of tetracycline systems or search our collection for all dox-regulated plasmids.
| ID | Plasmid | Description | PI | |
|---|---|---|---|---|
| 63800 | PB-TRE-dCas9-VPR | Tet-inducible expression of SP-dCas9-VPR transcriptional activator with Tet-On 3G | George Church | |
| 87360 | TLCV2 | Lentiviral vector for tet-inducible Cas9-2A-EGFP expression. Based on LentiCRISPR v2. | Adam Karpf | |
| 167935 | pLenti-tetON-KRAB-dCas9-DHFR-EF1a-TagRFP-2A-tet3G | Tet-inducible expression of KRAB-dCas9-DHFR for CRISPRi. See Plasmid #167937 for CRISPRa | Emma Rawlins | |
| 72835 | pMK243 (Tet-OsTIR1-PURO) | Expresses OsTIR1 under the control of a TRE3GS promoter for conditional auxin-inducible degron system | Masato Kanemaki | |
| 92099 | AAVS1_Puro_Tet3G_3xFLAG_Twin_Strep | Tet-inducible expression gene of interest fused with 3xFLAG and 2xStrep-tag from TRE3G promoter for Tandem-Affinity Purification. | Yannick Doyon | |
| 20321 | TetO-FUW-OSKM | Tet-On inducible expression of mouse Oct4, Sox2, Klf4, and Myc for iPS cell generation | Rudolf Jaenisch | |
| 51543 | FUW-tetO-hOKMS | Tet-On inducible expression of human Oct4, Sox2, Klf4, and Myc for iPS cell generation | Tarjei Mikkelsen | |
| 172115 | PB-TO-hNGN2 | Piggybac Tet-On plasmid for differentiating iPSCs into glutamatergic neurons via human NGN2 expression. See more from the iPSC Neurodegenerative Disease Initiative (iNDI) Collection. | Michael Ward | |
| 105840 | pUCM-AAVS1-TO-hNGN2 | Introduction of dox-inducible human NGN2 into AAVS1 safe harbor site, for iPSC differentiation into cortical neurons | Michael Ward | |
| 100798 | pAAV-syn-FLEX-splitTVA-EGFP-tTA | Helper virus for monosynaptic tracing. To be coinjected with pAAV-TREtight-mTagBFP2-B19G (Plasmid #100799). | Ian Wickersham | |
| 172878 | pSF3-ultraID | Expression of UltraID in mammalian cells with the Tet-On or Tet-Off system | Julien Béthune | |
| 154939 | dCas13d-dsRBD-APEX2 | RNA-protein interaction mapping via Cas13-based APEX targeting | Alice Ting | |
| 85040 | pK170.AAV-TRE-Cre-WPRE (Supernova) | AAV vector for dox-inducible expression of Cre recombinase. | Takuji Iwasato | |
| 63704 | pRetroX GFP T2A Cre | Retroviral vector for dox-inducible expression of GFP T2A Cre recombinase. | Floris Foijer | |
| 165457 | AAVS1-Neo-TRE-CMV-Cre-rtTA | Dox-inducible expression of Cre recombinase. | Madeline Lancaster | |
| 198752 | DiLiCre 2.0 | Dox-inducible expression of a light-activated Cre recombinase. | Jacco van Rheenen | |
| 104102–104112 | TetBow | Series of plasmids for stochastic multicolor labeling for discriminating between neurons. | Takeshi Imai | |
| 155257 | Watermelon Pooled Library | Lentiviral barcode library for simultaneous tracing of clonal lineages and transcriptional and proliferative state of each cell in a population. Proliferation history is monitored by the dilution of a dox-inducible H2B-mCherry marker. | Aviv Regev, Joan Brugge |
Additional Resources
References
Das, A. T., Tenenbaum, L., & Berkhout, B. (2016). Tet-On Systems for Doxycycline-inducible Gene Expression. Curr Gene Ther, 16(3), 156–167. https://doi.org/10.2174/1566523216666160524144041 (Link opens in a new window) PMID: 27216914 (Link opens in a new window)
Gossen, M., & Bujard, H. (1992). Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc Natl Acad Sci U S A, 89(12), 5547–5551. https://doi.org/10.1073/pnas.89.12.5547 (Link opens in a new window) PMID: 1319065 (Link opens in a new window)
Gossen, M., Freundlieb, S., Bender, G., Müller, G., Hillen, W., & Bujard, H. (1995). Transcriptional activation by tetracyclines in mammalian cells. Science, 268(5218), 1766–1769. https://doi.org/10.1126/science.7792603 (Link opens in a new window) PMID: 7792603 (Link opens in a new window)
Heinz, N., Schambach, A., Galla, M., Maetzig, T., Baum, C., Loew, R., & Schiedlmeier, B. (2011). Retroviral and transposon-based tet-regulated all-in-one vectors with reduced background expression and improved dynamic range. Hum Gene Ther, 22(2), 166–176. https://doi.org/10.1089/hum.2010.099 (Link opens in a new window) PMID: 20825282 (Link opens in a new window)
Krueger, C., Pfleiderer, K., Hillen, W., & Berens, C. (2004). Tetracycline derivatives: alternative effectors for Tet transregulators. BioTechniques, 37(4), 546–550. https://doi.org/10.2144/04374BM04 (Link opens in a new window) PMID: 15517964 (Link opens in a new window)
Loew, R., Heinz, N., Hampf, M., Bujard, H., & Gossen, M. (2010). Improved Tet-responsive promoters with minimized background expression. BMC Biotechnol, 10, 81. https://doi.org/10.1186/1472-6750-10-81 (Link opens in a new window) PMID: 21106052 (Link opens in a new window)
Resnitzky, D., Gossen, M., Bujard, H., & Reed, S. I. (1994). Acceleration of the G1/S phase transition by expression of cyclins D1 and E with an inducible system. Mol Cell Biol, 14(3), 1669–1679. https://doi.org/10.1128/mcb.14.3.1669-1679.1994 (Link opens in a new window) PMID: 8114703 (Link opens in a new window)
Urlinger, S., Baron, U., Thellmann, M., Hasan, M. T., Bujard, H., & Hillen, W. (2000). Exploring the sequence space for tetracycline-dependent transcriptional activators: novel mutations yield expanded range and sensitivity. Proc Natl Acad Sci U S A, 97(14), 7963–7968. https://doi.org/10.1073/pnas.130192197 (Link opens in a new window) PMID: 10859354 (Link opens in a new window)
Zhou, X., Vink, M., Klaver, B., Berkhout, B., & Das, A. T. (2006). Optimization of the Tet-On system for regulated gene expression through viral evolution. Gene Ther, 13(19), 1382–1390. https://doi.org/10.1038/sj.gt.3302780 (Link opens in a new window) PMID: 16724096 (Link opens in a new window)
External Resources
Credits
- Contributing Authors
- Written and reviewed by the Scientific Curation team at Addgene.
- Last Updated
- Content last reviewed on 20 June 2025. Catalog items are updated more frequently.
Do you have suggestions for other plasmids that should be added to this list?
Fill out our Suggest a Plasmid form or e-mail [email protected] to help us improve this resource!



